|
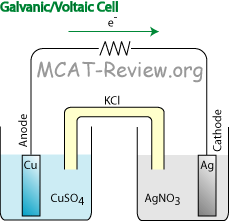
You find reduction potentials in a table:Ĭhlorine has high electron affinity, it loves to gain electrons and being reduced.Reduction half reaction decribes the species that gains electrons (decreases in charge).Oxidation half reaction describes the species that loses electrons (increases in charge).Oxidation is an increase in charge, Reduction is a decrease in charge.Oil Rig : Oxidation Is Losing e - Reduction Is Gaining e.This is because reduction occurs at the cathode to receive electrons. Electrons travel into the cathode, where it crashes into the cations on the surface of the cathode.Electrons shoot out of the anode because oxidation occurs there to lose electrons.electron flow oxidation, and reduction at the electrodes.For example, 1 mol of Cu is deposited for every 2 mols of electrons for the following half reaction: Cu 2+ + 2e - → Cu. Then using the half equation stoichiometry, you can find out how many mols of element is made for every e - transferred. Using this equation, you can solve for n, mols of electrons.Current x time = mols of e - x Faraday's constant.Faraday's constant = coulombs of charge per mol of electron = total charge over total mols of electrons.Current = coulombs of charge per second. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
